Category: Magnesium
Osteoporosis Medications, Action and Side Effects
May 25, 2019 3:40 pm
Treatment options should be approached is a global and systemic fashion. It is critical that the nutritional status is at its best possible and optimized for important healthy bone vitamins and minerals. Low protein needs to be corrected. Special attention should be given to nutrients, minerals and vitamins. These include Protein, Calcium, Magnesium, Vitamin D, and Vitamin K1/K2to name a few.
Healthy bones require ongoing and routine force in the form of exercise to remain health. Just as exercise improves muscle strength, it also improves bone health. Exercise is also critical in improving bone structure and density. Ideally, exercise should be weight bearing and resistance. Examples include: hiking, walking, jogging, climbing stairs, playing tennis, and dancing. Resistance type exercise is weight lifting and resistance bands. These exercise work by creating a pull or force on the bone either by gravity, movement or weight. Always check with your physician before beginning an exercise routine, start slowly and building up to longer periods of time. The ideal goal would be at least 30 minutes a day, every day, if you are able.
We frequently see patients immediately started on osteoporosis medications without checking or improving some of the nutritional markers noted above or without looking at exercise history. In some case, the medication recommended are contraindicated due to nutritional status.
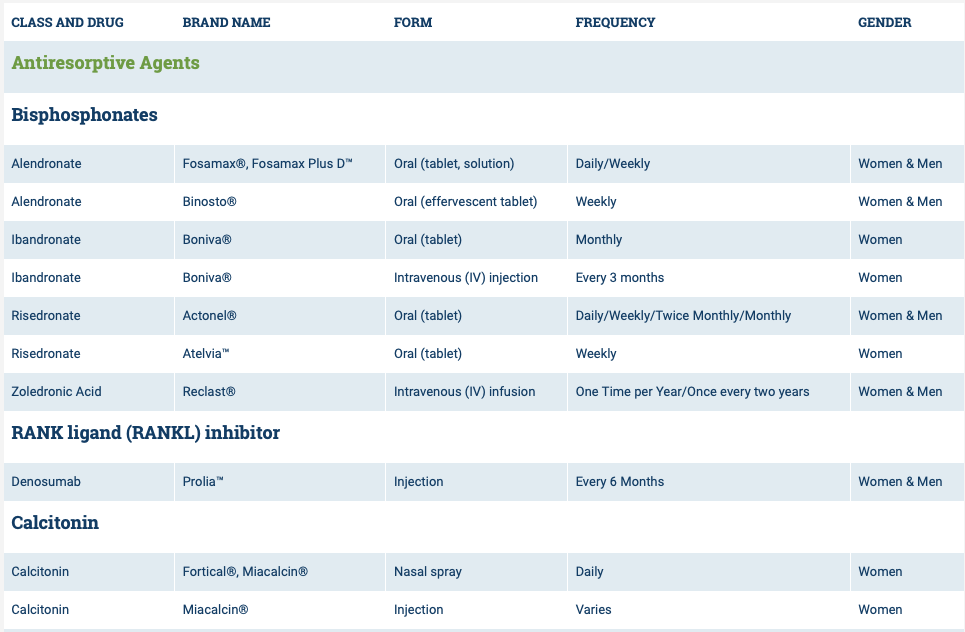
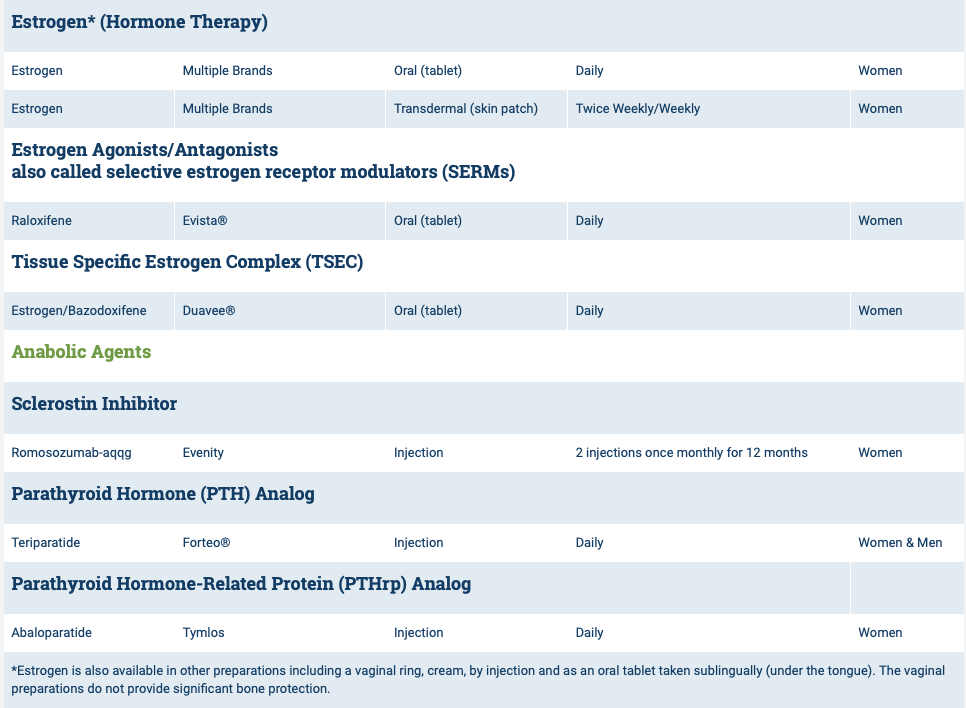
The medications can be grouped in to those that help with new bone formation (Anabolic agents) or those that help by suppressing the bone breakdown phase (Antiresorptive agents).
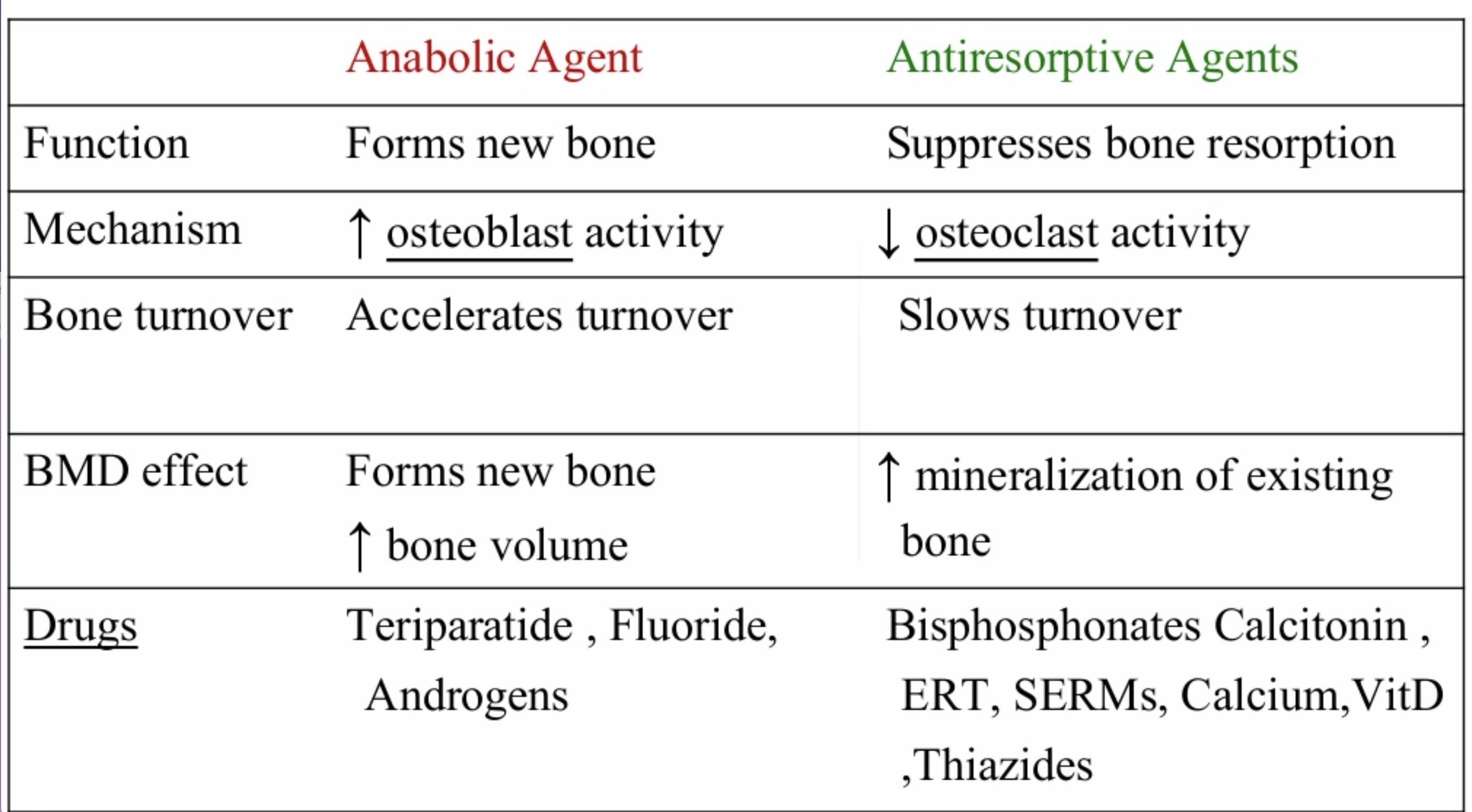
National Osteoporosis Foundation has an exhaustive list (below) of medications for treatment of Osteoporosis.
The table below outlines the side effects and mechanism of the actions of the common medications used for treatment of osteoporosis which was published by the University Health News Publication on August of 2014.
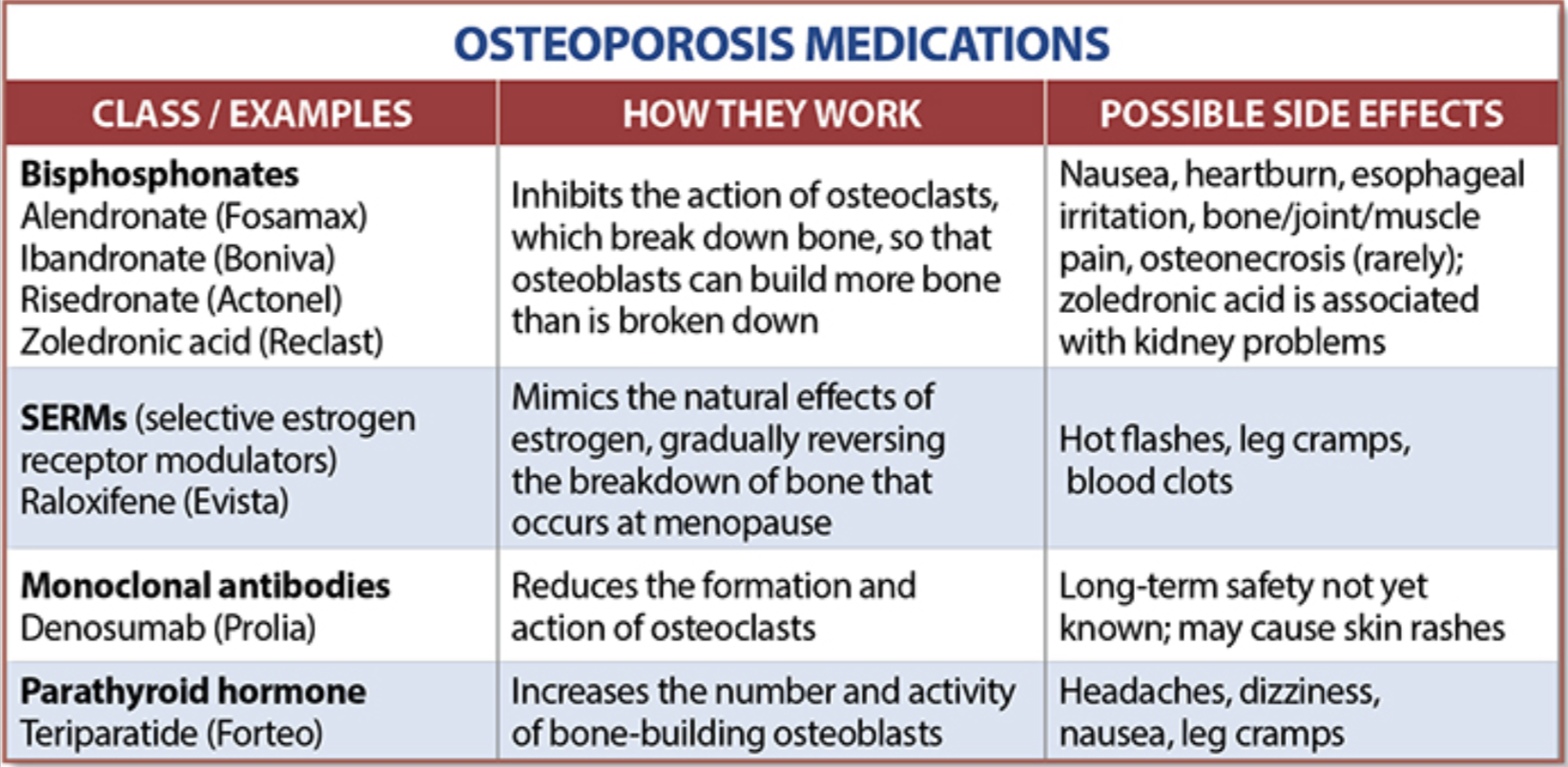
With all this information, the few points to remember is that the most important factors in healthy bone structure are the nutritional status Protein, Calcium, Magnesium, Vitamin D, and Vitamin K1 levels.
This is an animation of normal bone Metabolism. It shows how bone structures is taken down and rebuilt continuously. This allows for a healthy bone maintainence as we age. The key is the balance of breakdown (osteoclast) and the build up (osteoblast) activity is regulated. Osteoporosis develops when there is more breakdown that build up. 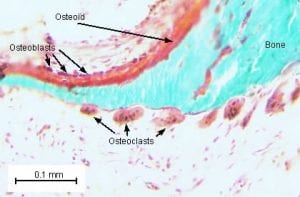
With permission of Dr. Susan Ott of University of Washington.
Additional information available on her site.
Past blogs on Bone Health.
Calcium Lab Results
June 05, 2018 3:26 pm
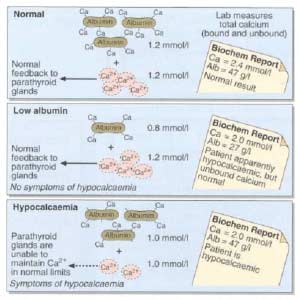
Calcium is measured to evaluate function and adequacy of a physiologic processes. Calcium plays a critical role in several body functions such as, coagulation pathways, bone health, nerve conduction, and other functions. It is important whenever you are evaluating laboratory results that you look at the whole picture of the person, including medications, other laboratory studies and health history. One value is not a stand alone result. There are many factors that effect calcium results.
Factors that effect calcium results: (not an all inclusive list)
pH
Albumin
Lactate
Heparin
Anticonvulsants
Renal Disease
Pancreatitis
The two most common issues following Weight loss Surgery or Duodenal Switch may be albumin level and Vitamin D level. Please see past blogs on Vitamin D. Magnesium may also play a role in a Duodenal Switch patient.
The most common calcium result drawn is the total calcium level. Laboratory results may not explicitly label it as such, however, it measures the calcium that is bound to protein. Ionized calcium is the free calcium that is representative of the true total calcium. Ionized Calcium can be measured by ordering specific lab. Alternatively, the Ionized calcium can be calculated by the following formula: Corrected calcium mg/dL = (0.8 * (Normal Albumin – Pt’s Albumin)) + Serum Ca ) or use the calculator at the bottom of this post.
The low Albumin level accounts for the low calcium level. This may be the reason for a patient with a low albumin/protein level, also having their calcium level reported as low. However, when adjusted for the protein deficiency the corrected calcium comes into normal range. Video of Trouseau’s sign of a patient with calcium deficiency.
The first step in a patient who has low calcium reported, is to make sure their protein and albumin levels are normal, along with Vitamin D.
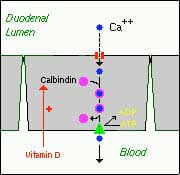
Calcium levels are managed by two processes major regularly hormones and influencing hormones. Controlling or major regulatory hormones include PTH, calcitonin, and vitamin D. In the kidney, vitamin D and PTH stimulate the activity of the epithelial calcium channel and the calcium-binding protein (ie, calbindin) to increase calcium absorption. Influencing hormones include thyroid hormones, growth hormone, and adrenal and gonadal steroids.
Further information on protein.
Further information on calcium.
Videos/Webinars on several of the above topics.
Corrected calcium = 0.8 * (4.0 – serum albumin) + serum calcium
Gas (Flatuence) GI upsetExclusive Member Content
July 27, 2017 7:32 am
Magnesium QuestionsExclusive Member Content
May 11, 2016 6:07 am
2016 Back on Track Giveaway!Exclusive Member Content
January 16, 2016 1:43 pm
Calcium – Revisited !Exclusive Member Content
October 05, 2015 8:06 am
Zinc InformationExclusive Member Content
April 17, 2015 7:22 am
Absorption of Minerals and Metals
January 12, 2009 6:42 am
The vast bulk of mineral absorption occurs in the small intestine. The best-studied mechanisms of absorption are clearly for calcium and iron, deficiencies of which are significant health problems throughout the world.
Minerals are clearly required for health, but most also are quite toxic when present at higher than normal concentrations. Thus, there is a physiologic challenge of supporting efficient but limited absorption. In many cases intestinal absorption is a key regulatory step in mineral homeostasis.
Calcium
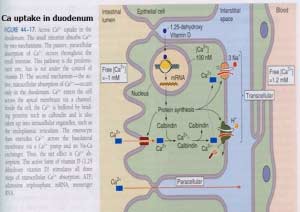
The quantity of calcium absorbed in the intestine is controlled by how much calcium has been in the diet during recent periods of time. Calcium is absorbed by two distinct mechanism, and their relative magnitude of importance is set by dietary calcium “history”:.
- Active, transcellular absorption occurs only in the duodenum when calcium intake has been low. This process involves import of calcium into the enterocyte, transport across the cell, and export into extracellular fluid and blood. Calcium enters the intestinal epithelial cells through voltage insensitive channels and is pumped out of the cell via a calcium- ATPase.The rate limiting step in transcellular calcium absorption is transport across the epithelial cell, which is greatly enhanced by the carrier protein calbindin, the synthesis of which is totally dependent on vitamin D.
- Passive, paracellular absorption occurs in the jejunum and ileum, and, to a much lesser extent, in the colon when dietary calcium levels have been moderate or high. In this case, ionized calcium diffuses through tight junctions into the basolateral spaces around enterocytes, and hence into blood. Such transport depends on having higher concentrations of free calcium in the intestinal lumen than in blood. Additional Calcium information here.
Phosphorus
Phosphorus is predominantly absorbed as inorganic phosphate in the upper small intestine. Phosphate is transported into the epithelial cells by contransport with sodium, and expression of this (or these) transporters is enhanced by vitamin D.
Iron
Iron homeostasis is regulated at the level of intestinal absorption, and it is important that adequate but not excessive quantities of iron be absorbed from the diet. Inadequate absorption can lead to iron-deficiency disorders such as anemia. On the other hand, excessive iron is toxic because mammals do not have a physiologic pathway for its elimination. Iron is absorbed by villus enterocytes in the proximal duodenum. Efficient absorption requires an acidic environment, and antacids or other conditions that interfere with gastric acid secretion can interfere with iron absorption.
Ferric iron (Fe+++) in the duodenal lumen is reduced to its ferrous form through the action of a brush border ferrireductase. Iron is the co-transported with a proton into the enterocyte via the divalent metal transporter DMT-1. This transporter is not specific for iron, and also transports many divalent metal ions.
Once inside the enterocyte, iron follows one of two major pathways. Which path is taken depends on a complex programming of the cell based on both dietary and systemic iron loads:
- Iron abundance states: iron within the enterocyte is trapped by incorporation into ferritin and hence, not transported into blood. When the enterocyte dies and is shed, this iron is lost.
- Iron limiting states: iron is exported out of the enterocyte via a transporter (ferroportin) located in the basolateral membrane. It then binds to the iron-carrier transferrin for transport throughout the body.
Iron in the form of heme, from ingestion of hemoglobin or myoglobin, is also readily absorbed. In this case, it appears that intact heme is take up by the small intestinal enterocyte by endocytosis. Once inside the enterocyte, iron is liberated and essentially follows the same pathway for export as absorbed inorganic iron. Some heme may be transported intact into the circulation. Additional Iron information here.
Copper
There appear to be two processes responsible for copper absorption – a rapid, low capacity system and a slower, high capacity system, which may be similar to the two processes seen with calcium absorption. Many of the molecular details of copper absorption remain to be elucidated. Inactivating mutations in the gene encoding an intracellular copper ATPase have been shown responsible for the failure of intestinal copper absorption in Menkes disease. A number of dietary factors have been shown to influence copper absorption. For example, excessive dietary intake of either zinc or olybdenum can induce secondary copper deficiency states. Additional Copper information here.
Zinc
Zinc homeostasis is largely regulated by its uptake and loss through the small intestine. Although a number of zinc transporters and binding proteins have been identified in villus epithelial cells, a detailed picture of the molecules involved in zinc absorption is not yet in hand. Intestinal excretion of zinc occurs via shedding of epithelial cells and in pancreatic and biliary secretions. A number of nutritional factors have been identified that modulate zinc absorption. Certain animal proteins in the diet enhance zinc absorption. Phytates from dietary plant material (including cereal grains, corn, rice) chelate zinc and inhibit its absorption. Subsistence on phytate-rich diets is thought responsible for a considerable fraction of human zinc deficiencies. Additional Zinc Information here