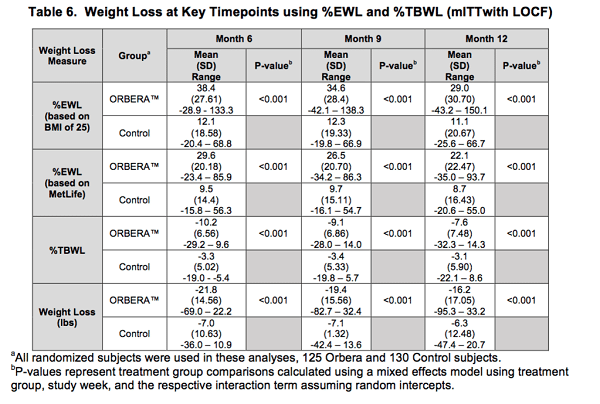
FDA approved the Orbera™ gastric balloon in 2015 for use and placement the United States. In this procedure, the surgeon performs an upper endoscopy, and the balloon is placed in the stomach and filled to the appropriate volume, which can range from 400-700 ml. The balloon has to be removed in 6 months. The outcome of balloon placement and removal is outlined by the manufacturer here. Please note that the results are only compared to diet and exercise and in that case at 12 months out (no long term studies available in US) has been described as it relates to diet and exercise as the base line. The patient who had the balloon placed, had initially lost 21.8 lbs. compared to 7.0 lbs. with diet and exercise at 6 months from the time of placement (when the balloon was removed). Leaving the balloon in for longer than 6 months, significantly increase the chanced of deflation and bowel obstruction. It is required that the balloons be removed 6 months after placement. At 9 months, the patient with the balloon removed had experienced weight gain where as diet and exercise group maintained the weight loss. This trend continued to 12 months. At 12 months, the balloon group had gained 26% of the weight loss ((21.8-16.2)/21.8=0.26, %26), where as the diet and exercise group had only gained 10% ((7.0-6.3)/7.0=0.1, %10). This means that at 12 months follow up, the balloon group gained weight back 3 times more than that of the diet and exercise group. It is to be noted that the initial weight loss for the balloon group was more than that of the diet and exercise group.
The indications for placement are very specific. BMI should be between 30-40 kg/m2, and the patient should be using the balloon “… in conjunction with a long-term supervised diet and behaviors modification program designed to increase the possibility of significant long-term weight loss and maintenance of that weight loss.”
The contraindications for the Orbera™ are listed on page 5 of the manufacturer published handbook by and include: previous weight loss surgery, presence of a gastric balloon (can not place a second one in if one is present) history of upper gastrointestinal diseases, including ulcers, reflux, hiatal hernia or other disorders. Pregnancy and breast feeding is also a contraindication for balloon placement.
In my opinion, there is no long-term data to support the use of this device unless the expectations for the patient are defined clearly. As the data suggests, this device will be ideal for patient who have some weight to loose and are able to adhere to a very well structured diet and exercise plan. Not to forget that the balloon has to be removed 6 months after the placement. According to the data, at the time, some weight gain is expected.
