Tag: Bowel obstruction
Biofilm
April 26, 2020 9:44 am
Written By: Eric Baghdasaryan, Maria Vardapetyan, Osheen Abnous
Biofilm are tiny microorganism-filled communities found throughout the human gastrointestinal tract and oral cavity. These communities adhere to both biological and non-biological surfaces within the human body. They provide their inhabitants with many competitive advantages that help these bacterial communities withstand biological, chemical, and physical stresses1. Within these communities, microorganisms collaborate with one another to increase the likelihood of growth and multiplication despite the harsh conditions of the human gut. Not all bacteria within these biofilms are harmful, but those that are go on to cause infections and other severe health problems for the host.
Biofilm formation
Studies have shown that approximately 60-80% of bacterial infections in the human body are associated with biofilm formation. Such findings have compelled researchers to investigate the complex factors responsible for biofilm formation.
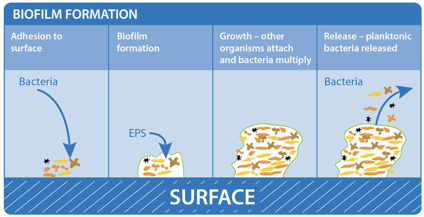
Figure 1: Biofilm adhesion, formation, and maturation. whiteley.com.au/biofilms
It was shown that bacteria anchor themselves to the mocosa surface. They collectively form a protective layer made of polysaccharides, proteins, and extracellular DNA. This forms the biofilm matrix. This biofilm matrix acts as a shield against antimicrobials, toxins, and antibodies. From here, biofilm associated bacteria go on to cause chronic infections characterized by persistent inflammation and tissue damage, initiating in the gut and adjacent regions of the gastrointestinal tract. Furthermore, some biofilm associated bacteria have shown the ability to disperse from this mature biofilm to colonize new niches, underscoring the association between local infections and systemic diseases such as atherosclerosis and rheumatoid arthritis2 caused by the buildup of biofilm dispersed bacteria and subsequent inflammation in the coronary vasculature and joint capsules, respectively. In fact, BADAS syndrome (bowel associated dermatosis-arthritis syndrome) is a condition where patients present themselves with small bumps on their inner skin (mainly along the vasculature) caused by the buildup of circulating microtoxins (very small clusters of bacteria). This may lead to chronic bacteremia in the bloodstream. Also, the over circulation of host immune complexes presents many problems for the patients, such as the development of arthritis and the accompanying joint pain, caused by the build up of white blood cells in host joint capsules. Patients often link the lumps to a dermatological condition, when in reality the condition is caused by a bacterial overgrowth originating in the gut and bowel. Clinicians now believe it is very likely these bacteria are biofilm-associated and a proper early diagnosis of the biofilm origin is critical to the prevention of BADAS and similar pathologies3.
The formation of biofilms has been studied on foreign substances such as intravenous catheters, orthopedic implants, and other biomaterials that have shown device-associated infections. However, it is commonly accepted that the majority of chronic bacterial infections involve biofilm formation on natural surfaces. The pathogenic bacterial overgrowth, forming the biofilms, have been linked to major diseases of the gastrointestinal tract including Inflammatory bowel disease and colo-rectal cancer4.
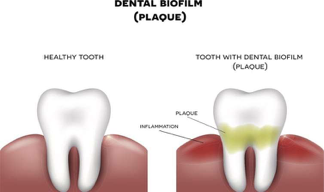
In addition to the gastrointestinal tract, biofilms can also be formed in the oral cavity. Over 700 bacterial species reside in the oral cavity. These contribute to the outgrowth of oral biofilms (otherwise known as dental plaque, see figure 2). These oral biofilms are responsible for major oral diseases such as tooth decay, gingivitis, and periodontitis. Moreover, those with periodontal infections have significantly increased risk of cardiovascular diseases, including atherosclerosis, myocardial infarction, and stroke2. The inflammation caused by oral biofilm may also be a contributing cause of conditions such as diabetes and rheumatoid arthritis1. Therefore, the control of oral biofilm growth before the development of oral infections is critical for the prevention of these system conditions.
Figure 2: Oral Biofilm Formation.
https://phys.org/news/2018-10-scientists-infection-causing-biofilms.html
This symbiotic (mutually beneficial) relationship between gut microbiota (bacteria living in our gut) and the host begins at birth and is crucial to our overall fitness and health. However, certain external and internal factors modify the gut microbiota. This causes the formation of a pathogenic biofilm, which leads to detrimental health conditions. The same bacteria that was once helping us by maintaining a healthy gastrointestinal tract, is now triggering disease conditions4.
Due to bacteria’s ability to translocate, migrate, and colonize new surfaces or niches, biofilm associated infections in the gut have been linked to systemic infections in other organs, including the joints, the skin, the eyes, the vasculature, the lungs, and even the central nervous system. It is assumed that the formation of a thick mucosal biofilm might be used as a diagnostic biomarker for the onset of systemic diseases. The outgrowth of a biofilm is widely viewed as the tipping point between two alternative states: a healthy and diseased gut1.
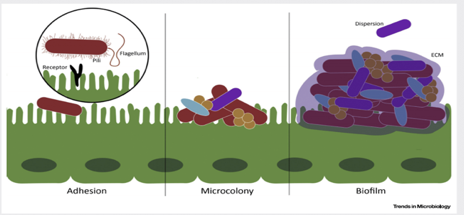
Figure 3. Biofilm matrix – a protective layer. Trends in Microbiology.
The biggest clinical challenge with biofilm-associated infections is their high resistance to antibiotic therapy. The effective therapeutic concentration of certain antibiotics to fight off bacteria within a biofilm (amount of the antibiotics needed in order to have positive therapeutic effects) is about 100-1000-fold higher than if the same bacteria were not associated with a biofilm2. The extracellular matrix, scaffold that keeps the bacteria anchored in place, prevents the penetration of host immune cells into the biofilm, thus contributing to the increased survival of the bacterial species living within the biofilm. Bacteria living within a biofilm also undergo an increased number of mutations, leading to the generation of more antibiotic-resistant phenotypes of bacteria. Finally, studies have shown that minimal concentrations of antibiotics may actually facilitate and stimulate biofilm formation, which can be extremely problematic in clinical treatment2. Therefore, to decrease the risk of biofilm induction, physicians should begin with very high doses of chemotherapeutics (antibiotics) from the very beginning of diagnosed infection. Looking ahead, there is clearly a need for novel biofilm-targeted therapies that are specifically made to prevent biofilm formation as well as eliminate the biofilm completely once it has already matured. Researchers have identified several drug candidates – DNase, lactoferrin, chlorhexidine, and taurolidine2 – that they believe have the potential to effectively penetrate and destroy components of the biofilm matrix. Further research is needed to determine their efficacy.
References
- Tytgat HLP, Nobrega FL, van der Oost J, de Vos WM. Bowel Biofilms: Tipping Points between a Healthy and Compromised Gut? Trends in Microbiology. January 2019;27(1): 17-25. doi:10.1016/j.tim.2018.08.009.
- Marcinkiewicz J, Strus M, Pasich E. Antibiotic Resistance: a “dark side” of biofilm-associated chronic infections. Polskie Archiwum Medycyny Wewnetrznej (Polish Archive of Internal Medicine). 2013;123(6):309-312.
- Buret AG, Motta JP, Allain T, Ferraz J, Wallace JL. Pathobiont release from dysbiotic gut microbiota biofilms in intestinal inflammatory diseases: a role for iron? Journal of Biomedical Science. January 2019;26(1) doi:10.1186/s12929-018-0495-4
Lymphatic Channels
May 22, 2018 8:21 am
We are all aware of the arterial and venous systems. Arteries take the oxygenated blood from the heart to the organs and the veins take the blood back to the lungs to unload the carbon dioxide and reload oxygen to be taken back to the organs. In addition to the arterial and venous vascular systems, we also have the lymphatic channels that flow into the lymphatic system.
The Lymphatic channels and system may be new to some, however, it is the third vascular network that is much less defined. The Lymphatic system collects fluids that has left the artierial/venous vascular system along their travel outlined above and take it back to the venous system. The lymphatic vessels transport this fluid to the lymph nodes throughout the body where the nodes filter the fluid of bacteria and harmful substances. Eventually, the fluid makes it way back to the venous system via the Superior Vena Cava. Additionally, Lymphatics collect the lipids within the GI tract and transport them to the venous system for metabolism. Most of the time these serosal lymphatic vessels are very small and hard to notice on the bowel.
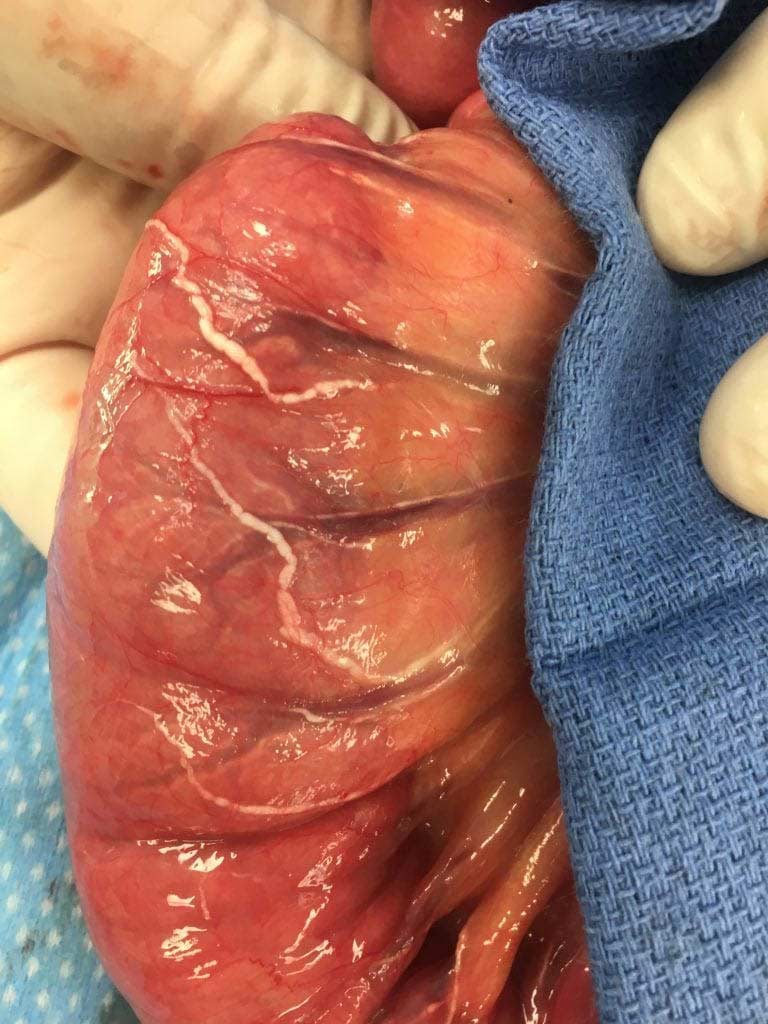
Example of Lymphatic channels
The following image is in a patient who had small bowel obstruction. The obstruction had resulted in vascular congestion at the base of the mesentery. The congestion had effected the low pressure system of the veins and the lymphatics disproportionately more that the arterial system. The white-milky tubular structures are the lymphatic channels filled with lipids.
There are three layers to the small intestinal lymphatic system, in the villi, submucosal and serosal layers and has the unique ability to transport absorbed intra-lumenal nutrients. There is a need for further research in the areas of health, obesity and disease in regards to the lymphatic system.
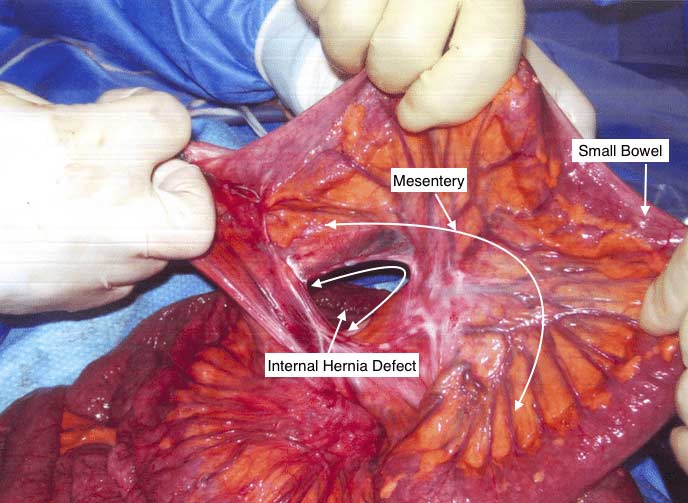
Internal Hernia And Bowel Obstruction
August 21, 2015 7:49 pm
Whenever there is a bowel resection with anastomosis made there will be a defect in the mesentery (the tissue that holds the blood supply and the nerves etc going to and from the bowel) that needs to be closed. In this particular case, the stitches that were used to close the defect were intact and yet the tissue had separated from it. The result is an internal hernia. This can cause bowel obstruction, where by a loop of the bowel can go through the defect and kink the bowel causing the blockage. In some cases, the internal hernia may reduce itself with intermittent symptoms of the bowel obstruction and in other cases it may require immediate emergent surgery. A CAT scan with oral and IV contrast is needed after Duodenal Switch to visualize the alimentary and bioliopancreatic limbs.
Symptoms may include but are not limited to:
- nausea
- vomiting
- abdominal bloating
- abdominal tenderness
- cramping abdominal pain
- diarrhea, constipation
- feeling of inability to completely empty bowels
- fever
- severe abdominal pain.
Bowel Obstruction After Duodenal Switch
August 19, 2014 4:47 pm
One of the potential complications of any abdominal surgery is Bowel Obstruction. If the treating physician (usually the primary care, or the emergency room doctor) is not absolutely clear of the anatomy of a patent post duodenal Switch or the Gastric bypass surgeries this will pose a diagnostic dilemma. In intact anatomy the GI tract start at the mouth and ends up at the rectum as a long tube. After the Duodenal Switch the small bowel has two parallel limbs, the alimentary limb brings the food down from the stomach, and the biliopancreatic limb brings down the biliopancreatic secretions. These two limbs join and form the common channel.
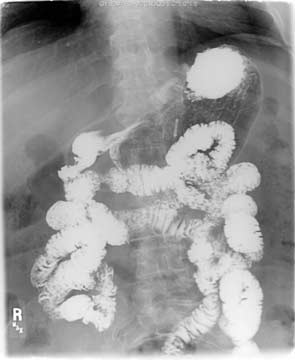
In normal anatomy, bowel obstruction may present with nausea, vomiting, abdominal cramps, inability to pass gas, and/or have bowel movements. In this case, the X-ray will also show dilated loops of bowel and if oral contrast is given with the X-ray, there will be no contrast past the obstruction. Think of it as a garden hose that has been kinked and no water is going thru.
In this upper GI- the contrast travels down the small bowel and the entire small bowel is the same caliber. This is normal study with no evidence of obstruction. In a patient with the DS, the patient my have the biliopancreaitc limb obstruction, with an identical X-ray as above, since the oral contrast given will never get to the biliopancreatic limb and it will not show if it is dilated or not.
In duodenal switch operation, a patient may have complete obstruction of the alimentary limb, with nausea, vomiting and abdominal pain, and yet have bowel movements because the content of the biliopancreatic limb is getting to the common channel. Similarly, a patient with biliopancreatic limb may have nausea, but no vomiting, because the obstructed biliopancreatic limb is not connected to the stomach and the content can’t not be expelled from the stomach.
The images of fluid filled loops of bowel are highly suspicious.
It is critical to make sure that a patient with a suspected bowel obstruction after the DS, is evaluated with the understanding that the common signs and symptoms, and the diagnostic workup will not provide an accurate picture. A patient with the DS or RNY, can have bowel obstruction and still have bowel movement, and no vomiting.
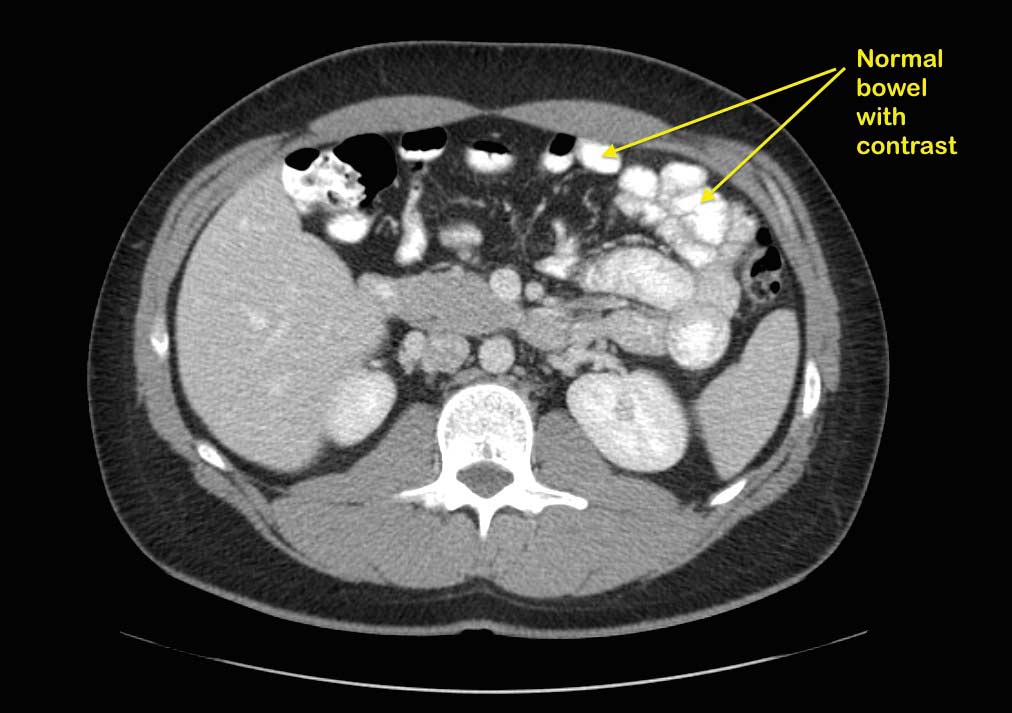
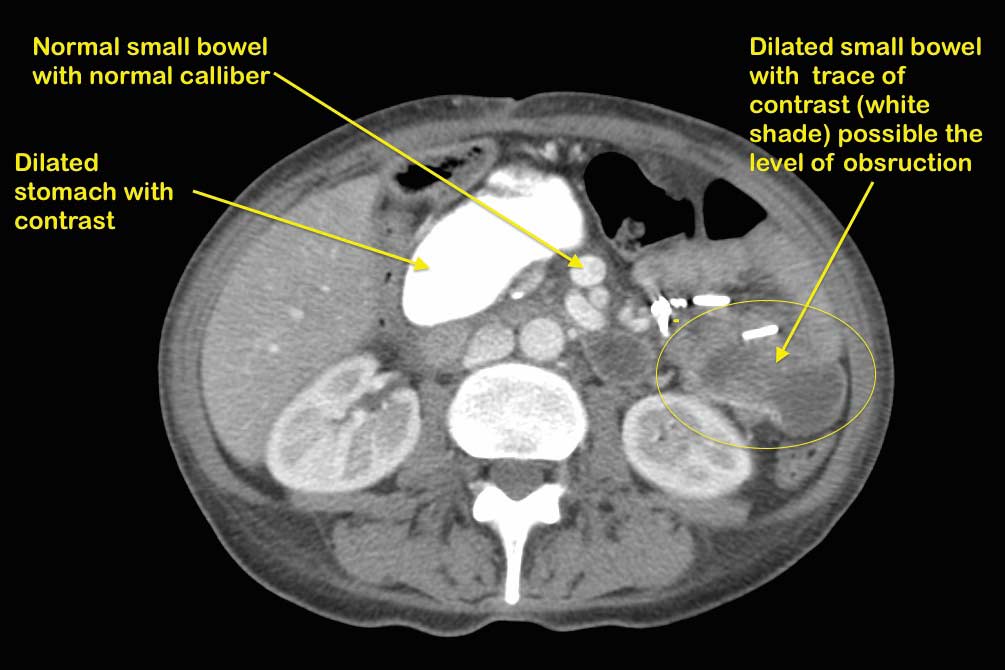
A patient with suspected bowel obstruction should have CT scan of the Abdomen with oral and IV contrast. The cardinal findings will be “dilated loops of bowel with no contrast within the lumen of the bowel”. This is highly suspicious for bowel obstruction after DS, where the regular x ray will not pick this up. Additionally, abnormal liver function test may suggest biliopancreatic limb obstruction.