This isn’t about which hospital is closest. Or who will pay for what. It’s about deciding which weight loss procedure is right for you, and finding the right surgeon to help you reach your goals.
When you’re ready to begin your journey, we invite you to start here. With Dr. Ara Keshishian and our top-notch team that blends advanced surgical treatments with true compassion to help you lose the weight. Start with the one on your shoulders.
To see what makes Central Valley Bariatrics the destination of choice for patients across the country and around the world, call today.



![]()
Keep our patients informed
At Central Valley Bariatrics, patients are the most important members of our team. A team dedicated to an individual and compassionate form of healthcare. Our goal is to keep every patient fully informed while providing the best quality care. We believe that continuous care is vital to the progress of each patient, decreases the risks of complications, and promotes excellent long-term success.
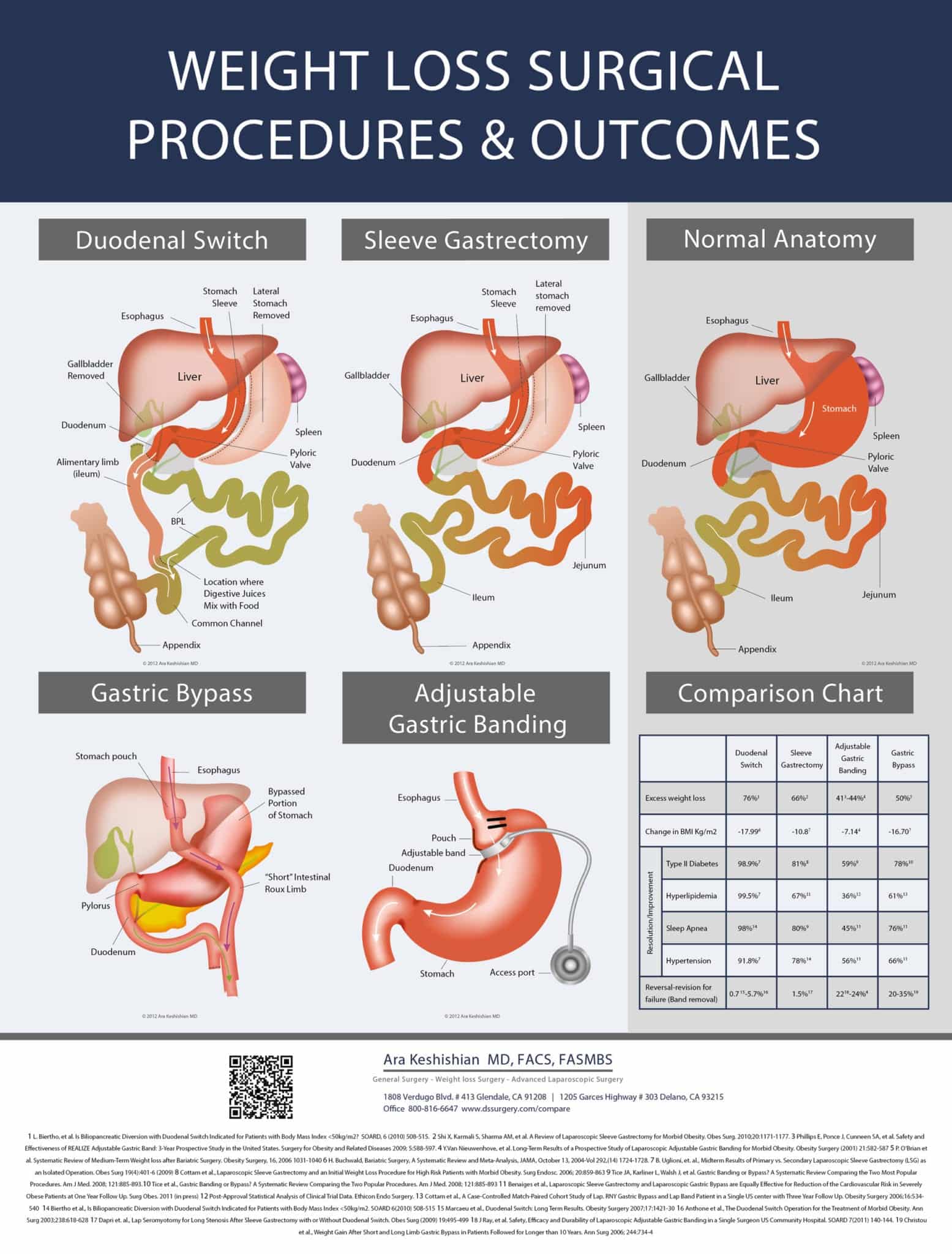
It is our mission to provide the finest and most current information, care, and available surgical treatment. We assist our patients in any way possible to achieve a positive medical outcome for their Duodenal Switch, Gastric Bypass Revisions, Revisions from other WLS, or Laparoscopic Sleeve Gastrectomy. We believe that the best patient-physician relationship can only be achieved through open communication and a mutual understanding with realistic expectations.





Dr. K gave me my life back. While I never reached my “ideal” weight, I lost over 100 pounds and have kept it off since 2002. To keep the weight off I do water aerobics 3 days a week. My friend and I started the water aerobics at our community pool 5 years ago. We go all year round and I teach one day a week. Thank you Dr. K.
Dr. Ara Keshishian is the most compassionate, caring and skilled physician I have ever met. He has a passion of helping people and has the talent and knowledge to actually accomplish just that. My experience with the entire staff was exemplary and I felt as though I was their only patient.
Dr. K is one of the hardest working & most caring individuals I have ever known. He is the very best at what he does & is fully committed to each & every one of his patients. He always answers my questions & makes sure that I fully understand what he is saying. My husband & I appreciate everything he has done for us…a life saver?…YES!… Thank you Dr. Keshishian.
Dr Keshishian saved my life. I know so many people say this but it’s true. I was morbidly obese and miserable. I couldn’t do any of the things I used to enjoy doing nor did I have the energy to do much of anything. When my primary doctor said I had to do something or die, I finally listened. He sent me to Dr Keshishian because he said he was the best. He was right. Dr Keshishian is so kind, patient and he listens. He truly has the heart of a teacher. He wants you to understand the procedures he uses. I can’t say enough goods things about this awesome man. If you’re considering WLS then you need to consider the best surgeon for the job and that’s Dr Keshishian.
This website has been prepared to help answer questions and concerns you may have about :
It is not intended to guide treatment, procedure, or substitute treatment by a trained healthcare professional.
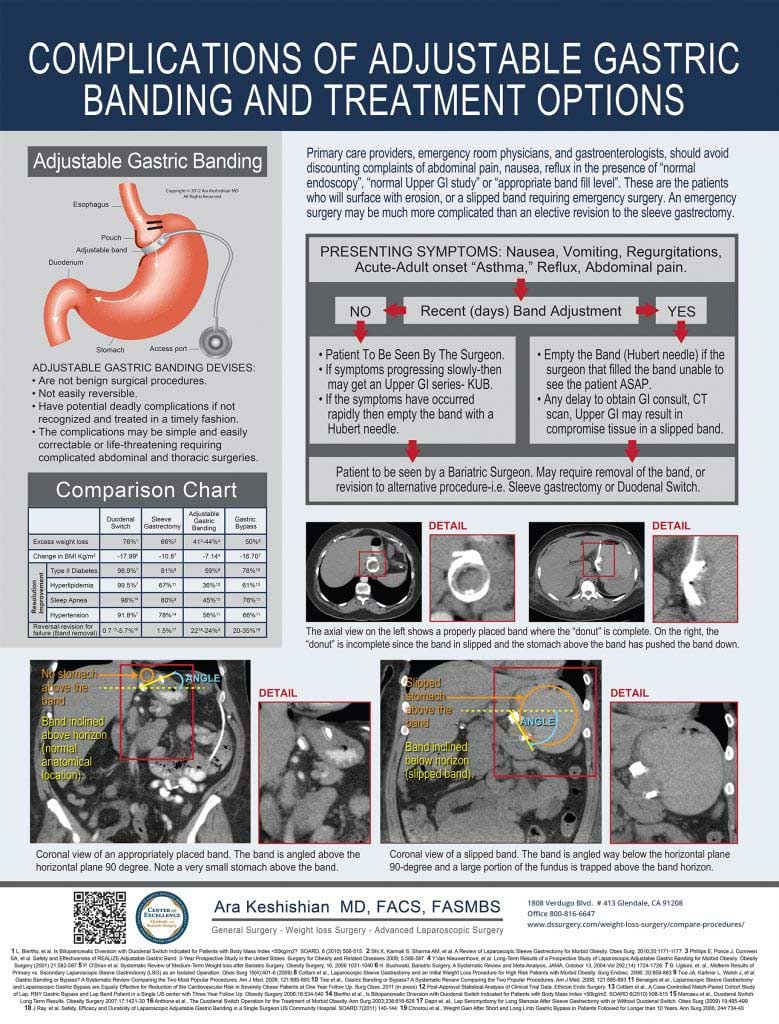
To ask further questions about Gastric Bypass alternatives, speak with a medical professional, obtain more specific information, or set up an appointment, please Contact Us or call 1-818-812-7222.
Error: Contact form not found.